 3.13 Total absorption spectrum of an idealized, productive (1mg chlorophyll a m~3) oceanic water, together with spectra of the individual absorbing components. The meaning of ABSORPTION SPECTRUM is an electromagnetic spectrum in which a decrease in intensity of radiation at specific wavelengths or ranges of. The two measured spectra can then be combined to determine the material's absorption spectrum. 3.13 shows, for an idealized, rather productive, ocean water, the individual absorption spectra of dissolved colour (gilvin), phytoplankton and detritus at plausible levels, and of water itself, together with the total absorption spectrum of the water due to all four components together.įig. The most straightforward approach to absorption spectroscopy is to generate radiation with a source, measure a reference spectrum of that radiation with a detector and then re-measure the sample spectrum after placing the material of interest in between the source and detector. To give an approximate indication of how the total absorption spectrum might be made up, Fig. This essay sketches the development of X-ray Absorption Fine Spectroscopy (XAFS) ever since the second half of 20th century. Absorption spectra are measured by varying the wavelength of the. Figure 3.12 shows the total absorption spectra of five Australian waters, three inland, one estuarine and one marine. Absorption spectra (also known as UV-Vis spectra, absorbance spectra and electronic spectra) show the change in absorbance of a sample as a function of the wavelength of incident light (Figure 1), and are measured using a spectrophotometer. The Jervis Bay water (e) contained 0.2mg phytoplankton chlorophyll a m~3 and was optically intermediate between Jerlov oceanic water types I and III.Ībsorption coefficient of pure water at that wavelength and the absorption coefficients due to dissolved and particulate colour, determined as described above. This is why hydrogen’s emission spectrum is the inverse of its absorption spectrum, with emission lines at 410 nm (violet), 434 nm (blue), 486 nm (blue. A free standing prism is placed directly in front of the projector and a cuvette slide is. When an electron drops down between levels, it emits photons with the same amount of energythe same wavelengththat it would need to absorb in order to move up between those same levels. Chlorophyll and turbidity data for waters a-d are given in the legends to Figs. Absorption Spectra A slide projector is used as a light source. 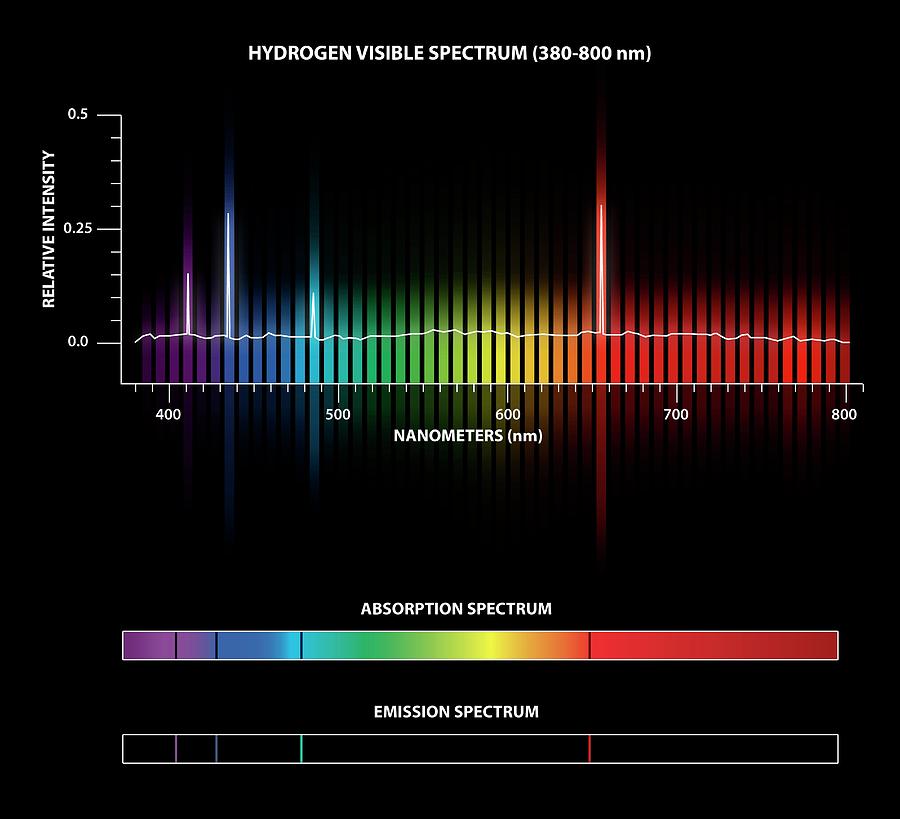
This causes an absorption spectra, as shown below. Waters a, b and c are inland, d is estuarine, e is marine (Tasman Sea). Absorption spectra When photons of light pass through a gas, the photons with the same energy as the energy gaps in the atoms can be absorbed. 3.12 Total absorption spectra of various natural waters in southeastern Australia (Kirk, 1981a and unpublished data). For any given water body, the total absorption coefficient at each wavelength is obtained by adding together the knownįig. The variation of this total absorption coefficient with wavelength is the absorption spectrum of the medium as a whole. 
At any wavelength, the aquatic medium has a total absorption coefficient, which is the sum of the absorption coefficients of all the light-absorbing components, at that wavelength. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
